Introduction
In general, the most of dosimeters are based on the physical and chemical effects in materials. Dosimeters based on the physical phenomena in the inorganic materials include Fluoro-glass dosimeter [1], optically stimulated luminescence dosimeter, electronic dosimeter. Dosimeters based on radiation-chemical reaction include alanine dosimeter [2], Cellulose triacetate (CTA) film dosimeter, Fricke dosimeter [3] and cerium dosimeter. Such dosimeters based on radiation chemistry are used for dosimetry in radiation processing relatively high dose region.
Moreover, other dosimetric methods include a biologic dosimeter. The conventional method of the biologic dosimeter is agarose gel electrophoresis [4] and comet analysis [5]. It is difficult for these methods to detect a conformation change of deoxyribonucleic acid (DNA). Those methods can be used only in high dose level. In addition, for strand break detection of the DNA, long running-time is needed. Other than these techniques, the unpaired electron in tooth enamel is evaluated by using electron spin resonance (ESR) [6ŌĆō8]. However, this method cannot estimate the structural change of DNA since only the analysis of the radicals is possible. Also, there is not a success of the measurement of bio-organic substance. Besides, X-band microwave used in ESR has a big dielectric loss by the water. Since the cell includes water, this characteristic leads big problem and difficulty in detection of formed radicals. Therefore, we have been developing a technique (Microwave Dielectric Absorption Spectroscopy) to detect structural change of DNA using the microwave of L-band. As for the L-band microwave dielectric absorption spectroscopy, influence by the dielectric loss of the water is smaller than that by the conventional X-band microwave dielectric absorption spectroscopy. This technique is different from the ultraviolet and visible photo-absorption spectroscopy to assay the amount of change based on Lambert-BeerŌĆÖs law. This method can estimate a change in the dielectric constant due to the orientation polarization in molecules as a shift of the resonance frequency. Therefore, it is expected that this method can measure the dielectric constant with high sensitivity.
The purpose of this research is to establish a new evaluation method of the damage of the DNA using microwave dielectric absorption spectroscopy. And we are aiming at the use in application to the biologic dosimeter, and the fundamental research of the biological effect of the radiation. Plasmid DNA was used as a biological molecule. The plasmid DNA is known to undergo structural change by damage caused by radiation. As the plasmid DNA has such a character, it is useful for biological research as materials. We thought a structural change of the plasmid DNA must be detectable using microwave.
In this paper, feature and construction of the system of the L-band microwave dielectric absorption spectroscopy and estimation of the DNA using the system are reported.
Materials and Methods
1. Samples
The sample is water, methanol and plasmid DNA solution. The dielectric constants of water and methanol are 78.54 and 24.74, respectively. The methanol solution was prepared with concentrations of methanol: 0, 3.12, 6.25, 12.5, 25, 50, and 100%.
The plasmid DNA pUC118 was multiplied by using Escherichia coli JM109 competent cells. Then the plasmid DNA was purified by QIAGEN Maxi kit (QIAGEN, Valencia, CA). The plasmid sample as a non-cutting was confirmed that more than 98% were supercoiled form by agarose gel electrophoresis. The plasmid DNA samples were prepared at concentrations of 13.1ŌĆō50.0 ╬╝g.cmŌłÆ3 in a 1├ŚTE buffer, pH 8.0. Linear form plasmid was prepared via double-strand break (DSB) by incubating supercoiled plasmid DNA with enzyme EcoR1 (TaKaRa, Tokyo, Japan) at a concentration of 15 U.mmŌłÆ3. Successful enzymatic treatment was confirmed by agarose gel electrophoresis. The concentrations of plasmid DNAs strand break products in the final solutions were 8.3ŌĆō49.0 ╬╝g.cmŌłÆ3.
2. Measurement
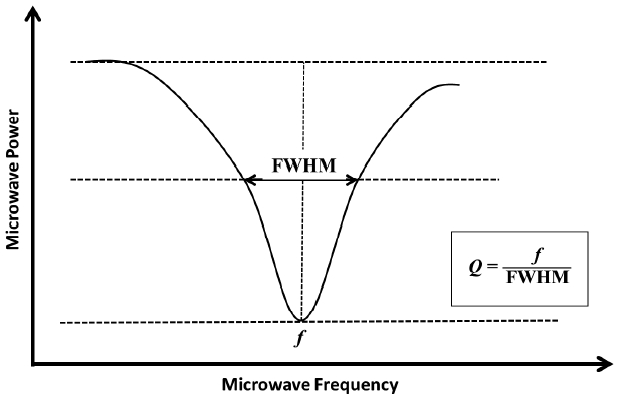
The sample solutions were put into the synthetic quartz cell with optical path length of 10.0 mm. It was installed in a cavity resonator using a measurement cell holder made of polyethylene. The resonance frequency and Q-value, shown in Figure 1, were measured by means of microwave dielectric absorption spectroscopy. The methanol-water mixed solution sample was measured at 21.5┬░C. The temperature was measured by digital temperature and humidity meter put outside of microwave cavity. Microwave dielectric absorption spectroscopy was carried out at 25.4┬░C and 25.2┬░C, in cases of supercoiled and linear form of plasmid DNAs, respectively.
3. Evaluation of dielectric constant change
The dielectric characteristics of the material can be expressed as Equation 1 [9].
where ╔øŌĆ▓ is real part of dielectric constant, ╔øŌĆ│ is imaginary part (dielectric loss) of dielectric constant. When a DNA molecular structure changed such as the DNA damages, ╔øŌĆ▓ and ╔øŌĆ│ change, resulting dielectric constant ╔ø changes in the cavity resonator. The change in the real part of the dielectric constant corresponds to the change in the resonance frequency. On the other hand, the change in the imaginary corresponds to the inverse Q-value. The dielectric constant changes with the molecular structure of the sample. The resonance frequency shifts to lower frequency when dielectric constant increases. The Q-value varies inversely with a half power width of a resonance frequency. These changes are measured by this method. Although all of the cavity resonators are not filled with a sample solution. When a sample is located at a center of the electric field, resonance curve of the frequency that existed as a standing wave is disturbed. As a result, the perturbation effect to perform a resonance with different frequency is adopted.
The microwave dielectric absorption spectroscopy system is composed of a cavity resonator, analog signal generator, circulator, power meter, and oscilloscope (Figure 2). Microwave oscillated from an analog signal generator is led to the inside of the cavity resonator through a circulator. The power of the reflected wave is measured by the power meter while sending reflected wave from the cavity resonator inside to the oscilloscope, and observing a waveform of the reflected wave. To measure a resonance frequency and power of the reflected wave automatically, an analog signal generator, oscilloscope, and power meter were controlled with a PC and newly developed software.
Results and Discussion
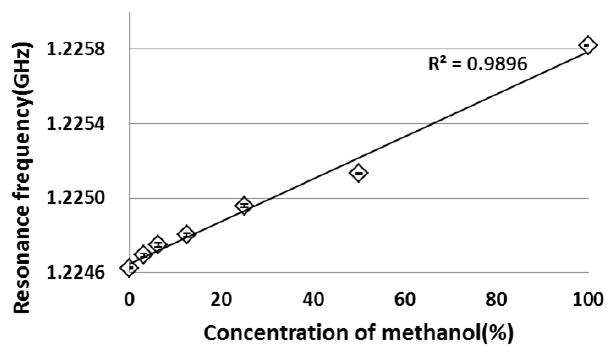
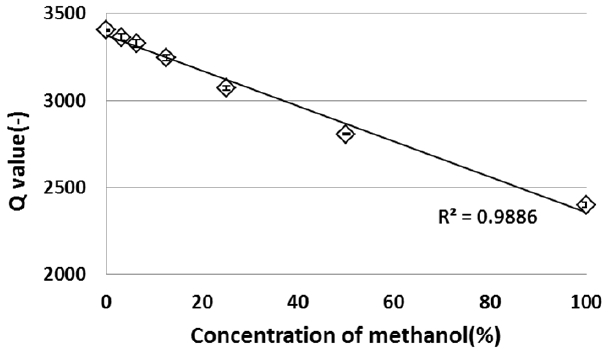
Results of measured resonance frequency and Q-value of the methanol-water solution are shown in Figures 3 and 4. The resonance frequency shifted to higher frequency with an increase in a concentration of methanol, and the Q-value decreased. The increase in a concentration of methanol corresponds to that in change of dielectric constants from water to methanol. The resonance frequency shifted to lower frequency with an increase of dielectric constant, and the Q-value increased. These results show that the change in the dielectric constant could be measured as a change in the resonance frequency and Q-value by the microwave dielectric absorption spectroscopy. This finding means that the measurement of organic molecules of the methanol is possible.
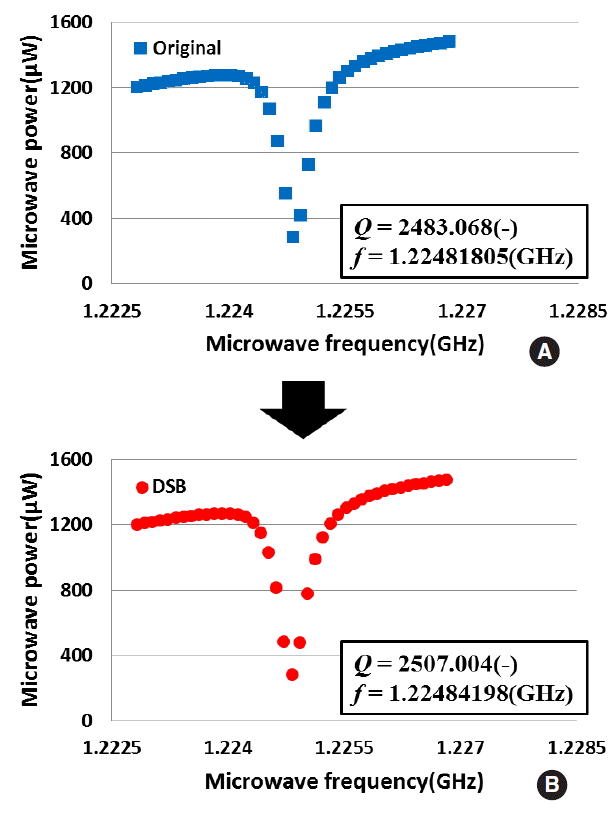
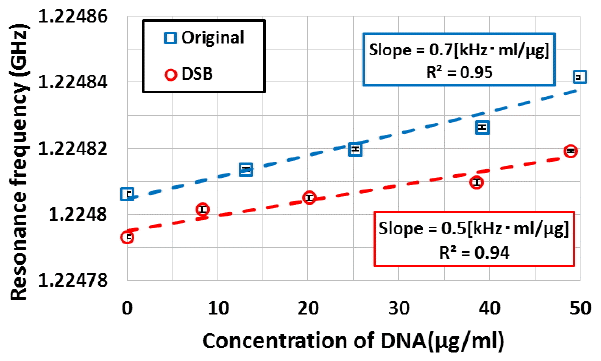
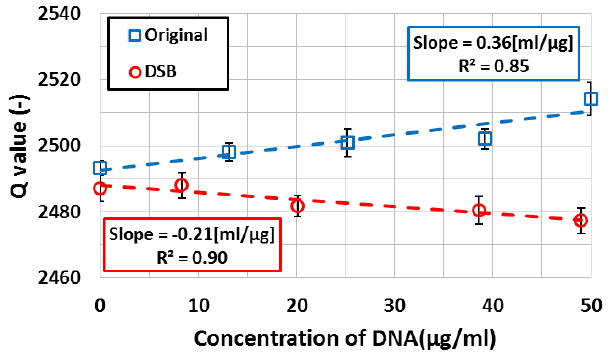
Concerning DNA macromolecule, we measured a dielectric constant change of DNA using the microwave dielectric absorption spectroscopy. Figure 5 shows the example of the microwave power spectra of DSB and non-damaged plasmid DNAs. The absorption peak in microwave power spectrum of the DSB plasmid DNA shifted from the non-damaged plasmid DNA. Moreover, the sharpness of absorption peak changed resulting in change in Q-value. Figure 6 shows that the results of resonance frequency of DSB and non-damaged plasmid DNAs. From this figure, we confirmed that a resonance frequency shifted to higher frequency with an increase in concentration of the plasmid DNA. The slope of the DSB plasmid DNA was estimated as about 0.5 kHz┬Ęcm3┬Ę╬╝gŌłÆ1. Also, the slope of the non-damaged plasmid DNA was estimated as 0.7 kHz┬Ęcm3┬Ę╬╝gŌłÆ1. The increasing resonance frequency tendency of DSB plasmid DNA is similar to non-damaged plasmid DNA. On the other hands, slope is different to each other. The difference of a slope of the resonance frequency is caused by the fact that plasmid DNA changed in structure from a supercoiled form to a linear form. Figure 7 shows the Q-values of DSB and non-damaged plasmid DNA. In the non-damaged plasmid DNA, the Q-value increased with an increase of concentration of DNA. However, the Q-value of the DSB plasmid DNA by enzymatic treatment decreased with an increase in concentration. The slope of the DSB plasmid DNA was estimated as about ŌłÆ0.21 cm3.╬╝gŌłÆ1. While the slope of the non-damaged plasmid DNA was estimated as 0.36 cm3.╬╝gŌłÆ1. The increasing Q-value tendency of DSB plasmid DNA is not similar to that of non-damaged plasmid DNA. These results suggest that the dielectric loss is increased by a change in the structure of plasmid DNA from supercoiled to linear. Therefore, the results show that microwave dielectric absorption spectroscopy is appropriate. Also, by using the calibration curve, it is expected that the proportion of each conformation of the mixed plasmid DNA can be obtained.
As mentioned in Introduction section [6ŌĆō8], ESR is powerful tool to analyze radiolytic formed radicals and one of the conventional techniques using microwave, however this method cannot estimate the structural change of DNA. One of the most important advantages of microwave dielectric absorption spectroscopy is that final products without unpaired electron can be analyzed.
In our previous paper, plasmid DNA irradiated with gamma-rays is measured using resonance frequency [10]. The resonance frequency shifted to lower frequency with an increase in absorbed dose. This result suggests that the real part of dielectric constant of plasmid DNA increases with an increase in damage of DNA by gamma-rays. On the other hand, in this paper, we estimated the structural change of plasmid DNA by using both of resonance frequency and Q-value. Moreover, if the structure of plasmid DNA changed from a supercoiled form (original) to a linear form (DSB), the Q-value changed more effectively than resonance frequency as shown in Figures 6 and 7. And by simultaneous measuring of resonance frequency and Q-value, the complex dielectric constant can be successfully obtained.
Therefore, we were succeeded in improving microwave dielectric absorption spectroscopy by employing a simultaneous measurement of resonance frequency and Q-value. This method will be applicable to the evaluation of the DNA damage caused by irradiation. We are planning to improve stability and accuracy of this method in the near future.
Conclusions
We developed the new evaluation method of DNA damage using microwave dielectric absorption spectroscopy. This system can estimate the difference in dielectric constant and structure of plasmid DNA. It is suggested that the microwave dielectric absorption spectroscopy can evaluate the quantity of DNA damage.